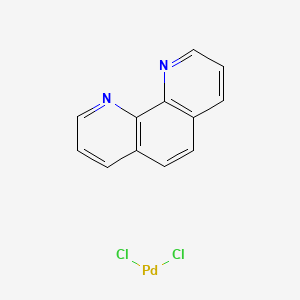
![1499115-56-8・Diphenyl Phenanthroline NNC Palladium [DPP-NNC Pd]・044-34351・040-34353[Detail Information] | [Synthesis & Materials]|Laboratory Chemicals-FUJIFILM Wako Chemicals U.S.A. Corporation 1499115-56-8・Diphenyl Phenanthroline NNC Palladium [DPP-NNC Pd]・044-34351・040-34353[Detail Information] | [Synthesis & Materials]|Laboratory Chemicals-FUJIFILM Wako Chemicals U.S.A. Corporation](https://labchem-wako.fujifilm.com/sc/01/1499115-56-8.png)
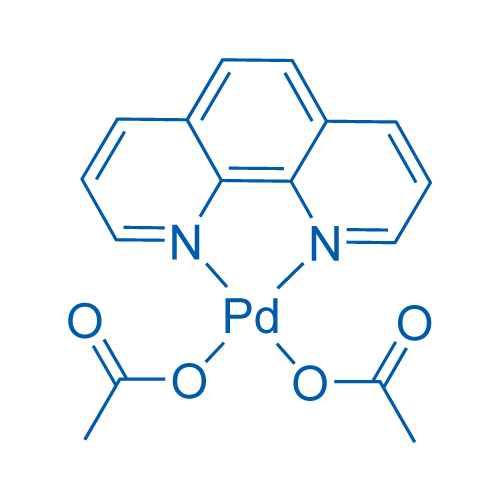
1499115-56-8・Diphenyl Phenanthroline NNC Palladium [DPP-NNC Pd]・044-34351・040-34353[Detail Information] | [Synthesis & Materials]|Laboratory Chemicals-FUJIFILM Wako Chemicals U.S.A. Corporation

2-Hydroxy-1,10-phenanthroline vs 1,10-Phenanthroline: Significant Ligand Acceleration Effects in the Palladium-Catalyzed Oxidative Heck Reaction of Arenes | Organic Letters
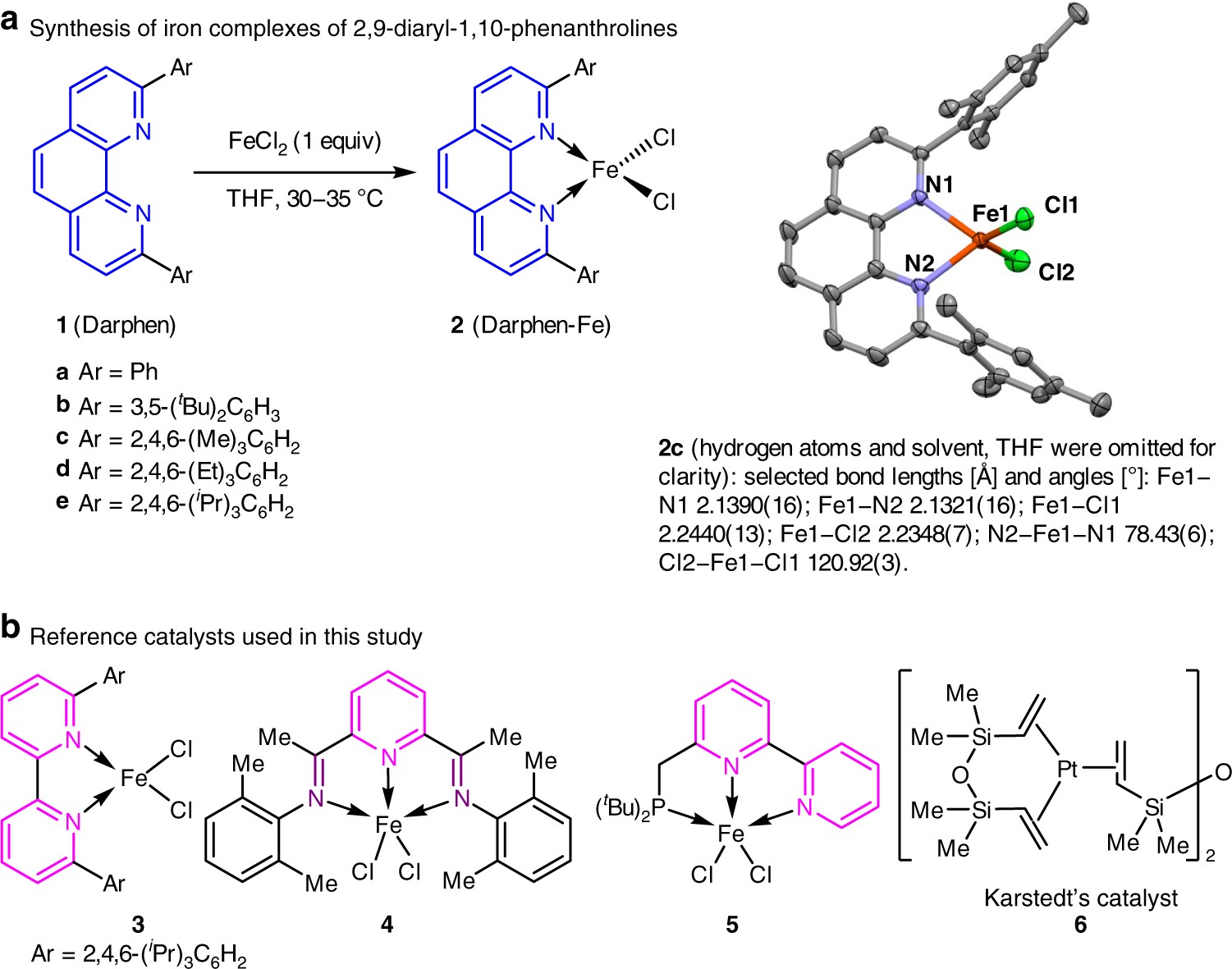
Ligands with 1,10-phenanthroline scaffold for highly regioselective iron-catalyzed alkene hydrosilylation | Nature Communications
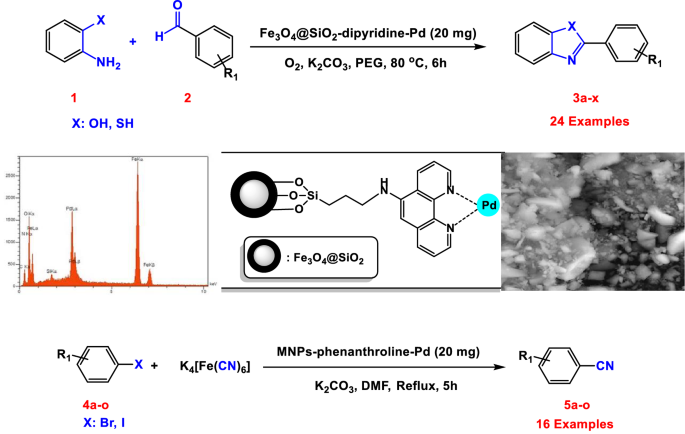
Palladium (II) Complex Supported on Magnetic Nanoparticles Modified with Phenanthroline: A Highly Active Reusable Nanocatalyst for the Synthesis of Benzoxazoles, Benzothiazoles and Cyanation of Aryl Halides | Catalysis Letters
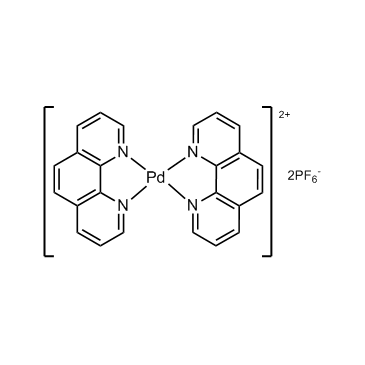
CAS 113173-22-1|Bis(1,10-phenanthroline)palladium(II) Bis(hexafluorophosphate) |buy 113173-22-1 |Inhibitor from supplier chemexpress.cn

SYNTHESIS AND STRUCTURE OF THE LINEAR BIMETALLIC ACETATE-PHENANTHROLINE COORDINATION POLYMER BASED ON PALLADIUM(II) AND NICKEL(II) IN A 2:1 METAL-TO-METAL RATIO | Journal of Structural Chemistry

US6281382B1 - Palladium phenanthroline acetate catalyst and a method of oxidizing side-chains of alkylbenzenes with catalyst - Google Patents

Highly active palladium catalysts containing a 1,10-phenanthroline analogue N-heterocyclic carbene for room temperature Suzuki-Miyaura coupling reactions of aryl chlorides with arylboronic acids in aqueous media - ScienceDirect
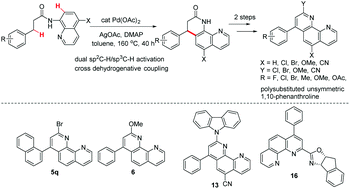
Practical synthesis of polysubstituted unsymmetric 1,10-phenanthrolines by palladium catalyzed intramolecular oxidative cross coupling of C(sp2)–H and C(sp3)–H bonds of carboxamides - Organic Chemistry Frontiers (RSC Publishing)

Synthesis, structures, photophysical properties, and catalytic characteristics of 2,9‐dimesityl‐1,10‐phenanthroline (dmesp) transition metal complexes - Cetin - 2020 - Journal of Polymer Science - Wiley Online Library


![Palladium Compounds [C-H Activation] | Tokyo Chemical Industry (India) Pvt. Ltd. Palladium Compounds [C-H Activation] | Tokyo Chemical Industry (India) Pvt. Ltd.](https://www.tcichemicals.com/medias/B5400.jpg?context=bWFzdGVyfHJvb3R8MzgzOTJ8aW1hZ2UvanBlZ3xoYzcvaDA5Lzg5MjkxOTk2NTI4OTQvQjU0MDAuanBnfDIwMDg1ZmRkYTE3ZTc1OTU4OWFmNDhhMzI3ZTEzODliNjI5MDBiZTUwODM1MTk1MTc3MGE0ZDQ4ZTk0ZmNiZTQ)